- Articles
Violence, mental illness, and the brain — A brief history of psychosurgery: Part 2 — From the limbic system and cingulotomy to deep brain stimulation
Abstract — Knowledge of neuroscience flourished during and in the wake of the era of frontal lobotomy, as a byproduct of psychosurgery in the late 1930s and 1940s, revealing fascinating neural pathways and neurophysiologic mechanisms of the limbic system for the formulation of emotions, memory, and human behavior. The creation of the Klüver‑Bucy syndrome in monkeys opened new horizons in the pursuit of knowledge in human behavior and neuropathology. In the 1950s specialized functional neurosurgery was developed in association with stereotactic neurosurgery; deep brain electrodes were implanted for more precise recording of brain electrical activity in the evaluation and treatment of intractable mental disorders, including schizophrenia, “pathologic aggression,” and psychomotor seizures in temporal lobe epilepsy. Psychosurgical procedures involved deep brain stimulation of the limbic system, as well as ablative procedures, such as cingulotomy and thalamotomy. The history of these developments up to the 21st century will continue in this three‑part essay‑editorial, exclusively researched and written for the readers of Surgical Neurology International (SNI).
ADVANCES IN NEUROSCIENCE AND THE KLÜVER–BUCY SYNDROME
One of the beneficial byproducts of frontal lobe surgery was the anatomic and physiologic knowledge that accumulated in the neurosciences from observations made both from animal experiments and human operations. This new information was added to what was already learned by the early neuroscientists. For example, in 1928 physiologist P.A. Bard had already noted that integrated rage reactions in cats could be elicited with diencephalic (hypothalamic) lesions.[4] Moreover, lobotomy studies revealed that ablative surgery of the orbitofrontal cortex resulted in damage to the limbic and autonomic system connections.[13] The prefrontal cortex was found to have direct thalamic‑cortical radiation connections involved in recruitment responses but also afferent and efferent connections to the hypothalamus modulating autonomic responses. Dr. Paul Yakovlev, aneuropathologist at Harvard, and associates studying white matter degeneration found that the cingulate gyrus was involved in lobotomy surgery regardless of the surgical approach. His work would soon lead to functional neurosurgery and cingulotomy and thalamotomy operations.[13,25]

In 1937, Heinrich Klüver (1897-1979), a German‑American experimental psychologist, investigated the drug mescaline and its effect on the brain of monkeys [Figure 1]. He asked American neuropathologist and later neurosurgeon, Paul Bucy (1904-1992) to perform temporal lobectomies in rhesus monkeys for his ongoing studies [Figure 2]. A fruitful collaboration ensued.[16] Decades later, Dr. Bucy was still active, becoming the founder (1972) and editor (until 1987) of the premier neurosurgical journal, Surgical Neurology, a parent publication to our online journal Surgical Neurology International.

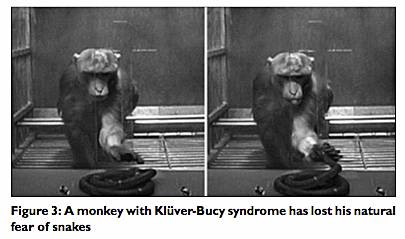
Drs. Klüver and Bucy reported that bilateral temporal lobectomies in monkeys resulted in a constellation of changes, including docility, hypersexuality, and hyperorality that was frequently associated with hyperphagia, and “psychic blindness.” This meant that the animals became tame with low levels of aggression, used inappropriate objects as sex objects and sexual stimulation, and examined objects by repeatedly placing them in their mouths. The animals also tended to overeat. Psychic blindness also meant that the animals suffered visual agnosia (despite intact vision), and a constellation of symptoms revolving around the lack of recognition of objects or their use culled from previous experience.[9,16,22] In their tameness and lack of recognition of objects, the monkeys would even lose their intuitive or conditioned fear of snakes [Figure 3].
In humans, the Klüver–Bucy syndrome manifests itself in patients who have suffered brain tumors or trauma affecting both of the temporal lobes, encephalitis, or carbon monoxide poisoning. And the clinical findings also include visual agnosia, memory impairment, and placidity sometimes associated with hyperorality.[9,22]
THE LIMBIC SYSTEM AND ITS ANATOMIC INTEGRATION INTO THE EMOTIONAL BRAIN

By 1949, the American physician, Dr. Paul D. MacLean (1913-2007), linked psychosomatic disease to what he called the “visceral brain.” He had connected clinical disease to physiologic disorders of the limbic system and the hypothalamus to the autonomic nervous system (ANS) and cerebral cortex.[17] He had based his findings on the work of another American physician and neuroscientist, Dr. James W. Papez (1883-1958), who in 1937 had described the limbic system as the anatomical‑physiological basis for human emotions [Figure 4].
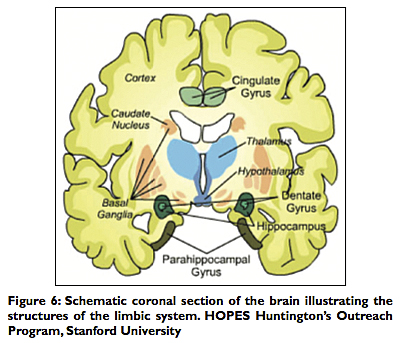
The neural circuit for the flow and expression of emotions was found to involve the sensory output of the thalamus to the sensory cortex, particularly the orbitofrontal cortex and associated cingulate gyrus. The neural pathway proceeds from the limbic cortex of the cingulate gyrus via the cingulum to the hippocampus, then via the fornix to the mammillary bodies and hypothalamus. The pathway thus completes a circuit, as the mammillothalamic tract sends neuronal impulses back to the anterior thalamus and the limbic cortex [Figures 5 and 6]. Perceptions and memories imbued with emotional content are then formed and stored. According to Dr. Papez, the mammalian brain evolved to respond to external threats with “fight or flight” reactions and to generate emotions and memories to these experiences.[6] Thus painful or pleasurable stimuli (and the emotional reactions they generate) are then embedded in the limbic circuit and the temporal lobes as useful memory that can be recalled as needed.[9,20,22]
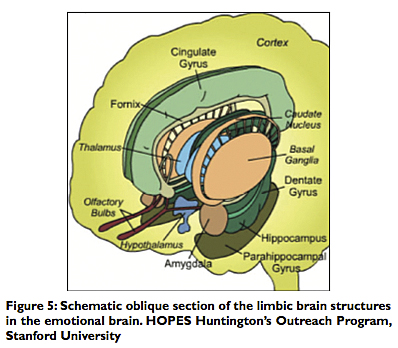
The limbic system (also referred to as “Papez circuit”) is phylogenetically derived from the primitive reptilian brain that permitted “fight or flight” reactions and allowed reptiles to adapt and survive for millions of years. It became the paleo‑mammalian brain integrated into the developing cerebral brain of mammals. In man it functions with our more complex and formidable cerebrum allowing for more complex emotional, social and sexual life, and the associated pleasurable or painful memories of those recollections. The limbic system consists of the following:
The hippocampus (the term derived from the ancient Greek for its shape of a “seahorse”) is located within the temporal lobe. The two hippocampi curve forward to reach their respective amydalas. They are required for the retention of short‑term into long‑term memory and cognition. Damage results in short‑term memory deficit, so that long‑term memory already acquired is spared, but new memories cannot be acquired or retained.[9,22] Classical clinical examples include Wernicke–Korsakoff syndrome as seen in alcoholics with thiamine deficiency and other confabulatory dementias.
The amygdalas are two almond‑shaped gray matter structures in the anterior temporal lobes set in front of each hippocampus. They flank the thalamus on both sides. The amygdala is involved in sending motivational signals related to fear, reward and punishment, and social, as well as sexual functions. It has also been associated with rage reactions and unprovoked aggression in humans with epileptiform foci detected by deeply implanted brain electrodes.[7,19]
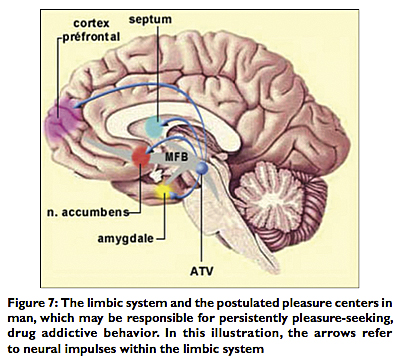
The fornix (the Latin term for “arch”) is a neural tract that conveys connections and signals from the hippocampus to the mammillary bodies and septal nuclei.[9] The mammillary bodies are important in the formulation of memory, while the septal nuclei, along with the nucleus accumbens, have been found by deep brain stimulation (DBS) studies in animals to be “pleasure centers.” The ventral tegmental area (VTA) of the midbrain sends signals via the medial forebrain bundle (MFB) to the nucleus accumbens, the septal nuclei and the amygdala, and via the cingulum to the prefrontal cortex using dopaminergic connections.[10,18] In man the chronic stimulation of the pleasure centers in the septal areas and nucleus accumbens is associated with persistent pleasure‑seeking behavior and drug addiction[7,22] [Figure 7].
The limbic lobes consist of the parahippocampal gyrus, the cingulate gyrus, and the dentate gyrus. These gray matter areas are part of the limbic system also involved in memory retention and retrieval, cognition, as well as more vegetative functions of the autonomic nervous system (ANS), such as maintenance of heart rate and blood pressure control. They are therefore intricately related to the hypothalamus, which, as we have noted, is also related to the limbic system. The orbitofrontal cortex is involved in attitudinal and motivational personality development and decision‑making ability, which is not surprising given what we have learned so far about loss of motivation, disinhibition, and the attainment of placidity resulting from frontal lobotomy in formerly aggressive, schizophrenic patients.
Other related areas of the limbic system are the basal ganglia, which are responsible for posture and modulating movement between the motor and associated cerebral cortex and the brainstem and cerebellum.[10]
FUNCTIONAL NEUROSURGERY VIA STEREOTACTIC CINGULOTOMY

The modern version of cingulotomy was developed in the early 1960s to replace frontal lobotomy, which had come into disrepute because of the excesses of Freeman and his followers in the 1950s.[21] Cingulotomy was developed by the neurosurgeons, Dr. Eldon L. Foltz and Dr. Lowell E. White, and subsequently modified by Dr. H. Thomas Ballantine (1912-1996) at Harvard Medical School, who added radiofrequency thermal coagulation and air ventriculography to the procedure [Figure 8]. Thus, by the 1970s cingulotomy involved stereotactic induction of radiofrequency lesions averaging 2 cm in length, designed to interrupt the cingulum, composed of association fibers between cortical and deeper limbic areas. The areas of thermocoagulation could include the anterior corpus callosum and the frontal lobe[2,3,12] [Figure 9].
Stereotactic cingulotomy was used more commonly for severe, intractable psychiatric disorders ranging from chronic pain syndromes (including thalamic pain), severe obsessive compulsive and panic disorders to agitated anxiety, psychotic depression, and schizophrenia. The most gratifying results were obtained in cases of depression and chronic pain disorders.
Criteria for psychosurgery, established by the National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research (1974-1978) to study and oversee psychosurgical procedures, were further delineated by the Massachusetts Institute of Technology (MIT). They required that patients to be evaluated for possible psychosurgery be referred by two psychiatrists, have available postoperative care, “be disabled by mental or physical anguish but have a premorbid history of effective functioning,” and have no previous relief from “appropriate treatments.”[12,24]
In 1980, JAMA contacted several neurosurgeons who had given up psychosurgical procedures because of political or social pressure. And yet, the officials for the National Commission for the Protection of Human Subjects were not opposed to the continuation of selected psychosurgical procedures. In fact, they found that 85 of the 137 patients studied suffered no permanent neurosurgical or behavioral deficits, and most patients had a slight improvement in intelligence testing postoperatively, probably because their severe preoperative symptoms had impaired their performance.[11,12]
Ballantine and co‑workers studied their series of patients and concluded that 75% were significantly improved with the most improvement noted in “unrelenting depression”; phobias were least amenable to surgical treatment, and three of these patients committed suicide. In one series of 686 patients who underwent stereotactic cingulotomy, complications were very few: Rarely seizures, one death, and two cases of hemiplegia.[3,5]
Other investigators found that cingulotomy makes psychiatric patients more responsive to psychotherapy and reduces untoward behavioral and autonomic hyperactivity that commonly result in therapy or management problems.[12] Indeed, lesions of the cingulum have been shown to reduce intestinal motility and improve lever‑pulling performance to avoid electric shock in 32 monkeys with experimentally created peptic ulcer disease. Curiously, experimentally induced morphine addiction was “attenuated” in 12 monkeys; and while active avoidance was improved, passive avoidance of an electric shock could not be learned by these monkeys.[12,14]
Nevertheless, by the 1980s, most neurosurgeons were reluctant to perform psychosurgery in the political climate that had begun in the 1960s and was pervasive at that time, a climate that prevails in the present day. By the 1980s, cingulotomy was the only psychosurgery procedure still performed, and Dr. Ballantine was one of the few surgeons who still performed it. He told JAMA in 1980 that in comparison to the United Kingdom, “We are impeded in this country by the climate of hysteria surrounding neurosurgery for psychiatric disorders.”[12]
PSYCHOSURGERY FOR AGGRESSIVE BEHAVIOR IN CHILDHOOD AND ADOLESCENCE
In the 1960s and 1970s functional neurosurgery, the ablation or stimulation of portions of the brain for the treatment of psychiatric or neurophysiological disorders was performed in different formats. In England, British psychosurgeons, for example, performed “limbic leucotomy” or subcaudate tractomy, sectioning of the ventromedial quadrant of the frontal lobe. And in 1975, forty years after the landmark World Congress of 1935 that brought frontal lobotomy into vogue, the Fourth World Congress of Psychiatric Surgery reported the new advances. Drs. Diekmann and R. Hassler of West Germany reported the successful treatment of violent male prisoners convicted of sex offenses. The selective lesions were done in the hypothalamus, which, with the pituitary gland, controls the endocrine system. Some of these felons had previously been referred for surgical castration but apparently the convicts opted for psychosurgery.[11,23]

In Japan and in the United States, uncontrollably aggressive and hyperactive children were also treated with psychosurgery in the 1950s and 1960s. Nevertheless, by the 1970s American and Japanese child psychiatrists reacted with horror to these procedures, and they were discontinued. In the U.S., Dr. Orlando Andy, Professor of Neurosurgery, Physiology and Biophysics at the University of Mississippi (Neurosurgery chairman, 1955-1979; d. 1997) was the most famous psychosurgeon to have operated on children [Figure 10]. He performed stereotactic thalamotomy in seriously incapacitated or severely hyperactive and aggressive children, who were difficult to manage by parents or institutions. He presented a series of 30 patients aged 6-49, including adults with Parkinson’s disease. All of the patients were shown to have abnormal brains by either radiographic or electrographic recording criteria. Dr. Andy surgically interrupted the thalamic neuronal circuits he believed were involved in these behavioral disorders. Most of his patients reportedly improved and were more manageable following surgery.
By the early 1970s, Dr. Andy had given up psychosurgery because of “sociologic pressure.”[1,11,19] Dr. Foltz, who had developed cingulotomy, also abandoned psychosurgery, despite the assurances of the National Commission, because of the fear of legal repercussions and malpractice lawsuits, not to mention the political and social climate that “made it so difficult to do cingulotomies that it didn’t seem worth the effort to battle that kind of attitude.”[11]
DEEP BRAIN STIMULATION (DBS) — CONTROL OF BEHAVIOR IN ANIMALS

The study of the electrical stimulation of the brain (ESB) is forever linked with the work of Dr. Jose Manuel Rodriguez Delgado (1915-2011) [Figure 11]. This relatively obscure scientist and prolific researcher has been an enigma, but over the years has almost become a cult figure in science, and his work has been described portentously as “mind control” experimentation. Dr. Jose Delgado was born in the province of Malaga in Spain. He obtained his M.D. degree from the University of Madrid and his Ph.D. from the Cajal Institute. Delgado had planned to follow in his father’s footsteps and become an ophthalmologist, but upon discovering the ground breaking work of his fellow countryman, Santiago Ramón y Cajal (1852-1934), the great Spanish neuropathologist and recipient of the Nobel Prize in Physiology or Medicine in 1906, Delgado decided to become a scientist.

In 1950 he accepted a position working under the famed neurophysiologist John Fulton at Yale, and for the next 20 years, Delgado conducted a monumental amount of research on ESB that reverberates to this day from neuroscience and sociology to science fiction. By the time, I entered college in 1970, he was already a legendary figure in the annals of DBS. In 1974, the Spanish minister of health enticed Delgado to return to Spain with his wife and children to organize a new medical school in Madrid [Figure 12]. Shortly before his death, he returned to America and died quietly in the United States in 2011.
Delgado’s work from the 1950s to the early 1970s was sometimes elegant, often rewarding, and always fascinating. Earlier investigators had studied “sham rage” in cats, but Delgado went much further. He investigated not only the ritualistic, motor component of sham rage, but also the more complex emotional and behavioral components of “true rage” in experimental animals and human subjects.[7,15]

Delgado found that “false” or sham rage could be evoked by stimulating the anterior hypothalamus of cats. The animal would respond with a generalized offensive display, not directed at other animals, but if a control animal in the cage retaliated, the stimulated cat would then lower its head and did not respond with the expected “fight or flight” reactions seen in nature [Figure 13].

When Delgado implanted the electrodes in the periventricular gray matter or even more specifically in the lateral hypothalamus of the cat, he could elicit true rage in which the experimental animal responded with the usual hissing, prowling, and then directly attacking the control cat. If the control cat retaliated, the stimulated cat would attack further with hissing and clawing [Figure 14]. The enraged animal became a bully in his cage, a situation analogous in humans to an individual with antisocial and violent behavior. In larger cages, Delgado observed, “the stimulated animal started prowling around looking for fights with other subordinated animals, but avoided the most powerful cat in the group. It was evident that ESB had created a state of increased aggressiveness, but it was also clear that the cat directed its hostility intelligently, choosing the enemy and the moment of attack, changing tactics, and adapting its movements to the motor reaction of its opponents.”[7]
ESB of the lateral hypothalamus could even result in the enraged animal attacking the friendly experimenter when he approached its cage. Cats could also express their dislike of ESB of certain brain areas by hissing, growling, and could even be motivated to learn instrumental tasks to stop the stimulation[7] [Figure 15].
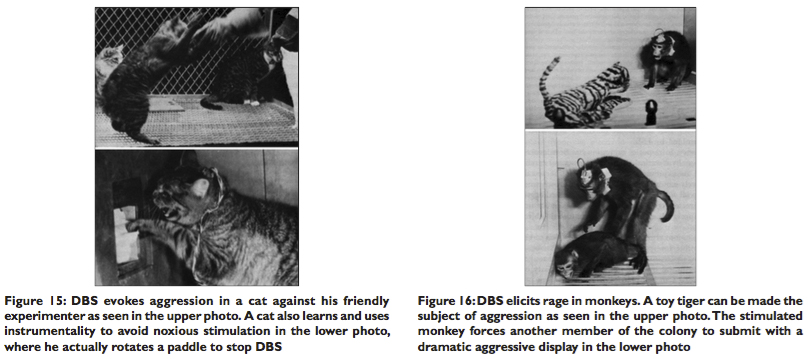
The hostility of these animals disappeared when the stimulation ceased, and the cats became once more friendly to other animals and to the experimenter. These experiments demonstrate that violent behavior and its associated emotional component could be modulated by ESB. Equally fascinating experiments in monkeys disclosed that DBS/ESB elicited complex and purposeful patterns of attack behavior influenced by their social organization, gender, sexual interactions, hierarchical status, and social dominance in the colony. A feline toy could even be made the focus of an attack [Figure 16]. Aggression was evoked when the thalamus and central gray matter were stimulated, and the aggression was directed and modified toward other members of the colony based on the status they held in the colony, the social rank of the stimulated monkey and the colony to which they belonged [Figure 17]. Once again this pattern of purposeful rage or aggression mimics the antisocial and violent behavior of some members of human society.

In Violence, mental illness, and the brain — A brief history of psychosurgery, Part 1[8] we discussed trephination as performed by the ancient shaman and the medieval surgeon. We also explained the circumstances that led to the recognition of the frontal lobe syndrome and the development of lobotomy to treat mental illness in the first half of the 20th century. In Part 3, the final installment of this article we will discuss Dr. Jose Delgado’s DBS studies in man and the controversial work conducted by Drs. Vernon H. Mark and Frank Ervin at Harvard, namely DBS with brain wave recordings and amygdalotomy in patients with intractable psychomotor seizures, as well as epilepsy and uncontrolled violence. Advances in neurologic research of violent individuals will be recounted, and the problem of uncontrolled rage and “pathologic aggression” in today’s “modern” society will be discussed from the sociologic as well as neurologic standpoint and ramifications.
REFERENCES
1. Andy O. Thalamotomy in hyperactive and aggressive behavior. Presented before the International Society of Research In Stereoencephalotomy. New York, September 27, 1969.
2. Ballantine HT Jr, Giriunas IE. Advances in psychiatric surgery. In: Rasmussen T, Marino R, editors. Functional neurosurgery. New York: Raven Press; 1979. p. 155‑64.
3. Ballantine HT Jr, Bouckoms AJ, Thomas EK, Giriunas IE. Treatment of psychiatric illness by stereotactic cingulotomy. Bio Psychiatry 1987;22:807‑19.
4. Bard PA. A diencephalic mechanism for the expression of rage with special reference to the sympathetic nervous system. Am J Physiol 1928;84:490‑515.
5. Cole HM. Stereotactic cingulotomy. JAMA 1985;254:2917‑818.
6. Dalgeish T. . Nature Neurosci 2004;5:582‑9. [Last accessed on 2013 Mar 01].
7. Delgado JM. . New York: Harper and Row; 1986. [Last accessed on 2013 Mar 01].
8. Faria MA. . Surg Neurol Int 2013;4:49. [Last accessed on 2013 May 01].
9. Fulton J. The limbic system. Yale J Biol Med 1953;26:107‑18.
10. Gilman S, Newman SW. Manter and Gatz’s Essentials of Clinical Neuroanatomy and Neurophysiology. 8th ed. Philadelphia: F. A. Davis Co.; 1992. p. 207‑61.
11. Gonzalez ER. Psychosurgery waiting to make a comeback? JAMA 1980;244:2245‑51.
12. Gonzalez ER. Treating the brain by cingulotomy. JAMA 1980;244:2141‑7.
13. Greenblatt M, Arnot R, Solomon HC. Studies in Lobotomy. New York: Grune and Stratton; 1950.
14. Hitchcock ER, Ballantine HT, Meyerson BA, editors. Modern Concepts in Psychiatric Surgery. New York: Elsevier/North Holland Biomedical Press; 1979. p. 111‑27.
15. Horgan J. . Sci Am 2005;293:22‑73. [Last accessed on 2013 Mar 1].
16. Klüver H, Bucy PC. Psychic blindness and other symptoms following bilateral temporal lobectomy in Rhesus monkeys. Am J Physiol 1937;119:352‑3.
17. MacLean PD. Psychosomatic disease and the “visceral brain”: Recent developments bearing on the Papez theory of emotion. Psychosom Med 1949;11:338‑53.
18. Margolis EB, Lock H, Hjelmstad GO, Fields HL. J Physiol 2006;577:907‑24. [Last accessed on 2013 Mar 1].
19. Mark VH, Ervin FR. Violence and the Brain. New York: Harper and Row; 1970. p. 1‑174.
20. Papez JW. A proposed mechanism for emotion. J Neuropsychiatry Clin Neurosci 1995;7:103‑12.
21. PBS. . Video‑documentary, January 21, 2008. [Last accessed on 2013 Feb 27].
22. Salloway S, Malloy P, Cummings JL, editors. The Neuropsychiatry of Limbic and Subcortical Disorders. American Psychiatric Pub, 1999.
23. Sweet WH, Obrador S, Martin‑Rodriguez JG, editors. Neurosurgical Treatment in Psychiatry, Pain, Epilepsy. Baltimore: University Park Press; 1977. p. 451‑62.
24. U.S. Public Health Service. Determination of the secretary regarding the recommendations on psychosurgery of the National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research. Fed Regist 1978;43:53242‑4.
25. Yakovlev PT. Motility, behavior and the brain; stereodynamic organization and neural coordinates of behavior. J Nerv Ment Dis 1948;107:313‑35.
Article written by: Dr. Miguel Faria
Miguel A. Faria, Jr., M.D. is Clinical Professor of Surgery (Neurosurgery, ret.) and Adjunct Professor of Medical History (ret.) Mercer University School of Medicine. He is an Associate Editor in Chief and a World Affairs Editor of Surgical Neurology International (SNI), and an Ex-member of the Injury Research Grant Review Committee of the Centers for Disease Control and Prevention (CDC). 2002-05; Former Editor-in-Chief of the Medical Sentinel (1996-2002), Editor Emeritus, the Association of American Physicians and Surgeons (AAPS); Author, ; Medical Warrior: Fighting Corporate Socialized Medicine (1997); and Cuba in Revolution: Escape From a Lost Paradise (2002).
This article was originally published in Surgical Neurology International and may be cited as: Faria MA. Violence, mental illness, and the brain – A brief history of psychosurgery: Part 2 — From the limbic system and cingulotomy to deep brain stimulation. Surg Neurol Int 2013;4(1):75. Available from: http://surgicalneurologyint.com/surgicalint_articles/violence-mental-illness-and-the-brain-a-brief-history-of-psychosurgery-part-2-from-the-limbic-system-and-cingulotomy-to-deep-brain-stimulation/
Copyright © 2013 Miguel A. Faria, Jr., MD